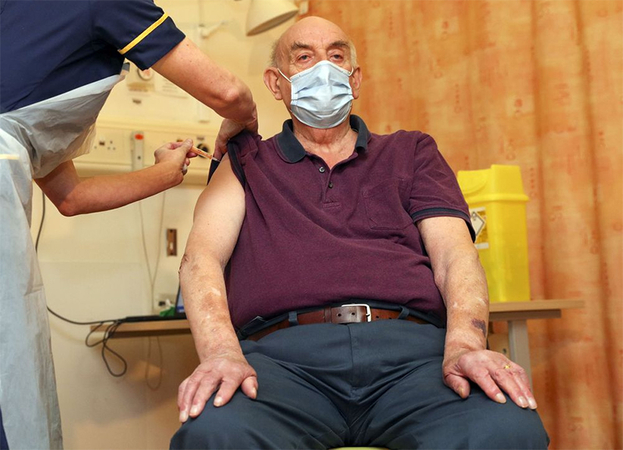
Britain gave the first shots of a coronavirus vaccine from AstraZeneca Plc and the University of Oxford, in a race against a faster-spreading coronavirus variant that's prompted new restrictions on movement across much of the country.
The first injection was administered Monday morning to an 82-year-old kidney dialysis patient at Oxford University Hospital.
Britain is stepping up its vaccine campaign as coronavirus infections surge across the country, putting the country on the brink of another national lockdown. It's moved more rapidly on approvals and rollouts than the U.S. or the European Union, clearing the AstraZeneca-Oxford product despite clinical trials that involved a smaller number of participants and that were complicated by a dosing error. A regulatory decision to lengthen the interval between doses of the two-shot vaccines to as many as 12 weeks has prompted further questions.
A new strain that's estimated to be as much as 70% more transmissible is fueling the pandemic's resurgence in the U.K. Schools have been closed across much of the country, and Prime Minister Boris Johnson has warned that tougher restrictions may be on the way.
U.K. regulators cleared the AstraZeneca-Oxford shot last week, marking its first approval worldwide. It's the second injection to be authorized for emergency use in Britain, after one from Pfizer Inc. and BioNTech SE received the go-ahead in early December.
More than a million people in Britain have received injections of the Pfizer-BioNTech vaccine, according to the Department of Health and Social Care.
The U.K. has raced ahead of France, where only about 500 people had got initial shots as of this weekend. President Emmanuel Macron has come under fire for the slow pace of that country's rollout, which has been hampered by caution amid high levels of vaccine skepticism. France is behind not just European neighbors such as Britain and Germany, but also Israel, where more than 12% of the population has already received injections.
In the U.K., more than 500,000 AstraZeneca-Oxford doses will be available as of Monday, and they will be delivered at hospitals for the first few days. The U.K. aims to expand the number of vaccination sites to more than 1,000, with as many as 100 more hospitals and 180 general practitioner-led services coming online this week.
Although the newly approved vaccine has shown lower effectiveness than the Pfizer-BioNTech one in clinical trials, it has some key advantages: It's cheaper and easier to transport and store, requiring only refrigerator temperatures rather than deep freezing. That makes it crucial for the broader global vaccination push.
David Nabarro, World Health Organization special envoy for covid-19, called for caution amid the global rush to vaccinate populations.
"I really would like to appeal to every leader just to really slow down a bit on the rush to get the vaccine into as many arms as possible and put more time into systematic planning on what has to be a really effective global operation," he said in an interview with Bloomberg Television.
The NHS is administering the first injections under a two-shot regime approved by the Medicines and Healthcare Products Regulatory Agency. The second can be given as many as 12 weeks later, as the U.K. seeks to maximize the number of vulnerable people who receive the first portion, which provides some protection from infection.
The U.K. has also taken a more flexible approach to the two-dose regimen, saying that in certain circumstances -- such as when it's not known which vaccine a patient received the first time around -- the second shot can be administered with a different company's product.
The U.K. regulator has yet to publish full data backing its decision to allow a greater time interval between shots, which has drawn opposition from the British Medical Association.
"It is grossly and patently unfair to tens of thousands of our most at-risk patients to now try to reschedule their appointments," said Richard Vautrey, chairman of the association's General Practitioners Committee.

