WASHINGTON - The National Institutes of Health on Friday removed restrictions that the Trump administration imposed on research using fetal tissue, allowing university researchers and government scientists freer rein to use material from elective abortions when studying diseases and possible treatments.
A brief update for outside scientists from the NIH director's office said the Department of Health and Human Services was reversing a 2019 decision that had required applicants for federal grants and contracts involving fetal tissue to undergo an extra layer of review by an ethics advisory board.
In a separate notice emailed Friday, NIH told its internal scientific and clinical directors that it was lifting a Trump-era ban on using federal money to buy human fetal tissue for biomedical studies by government employees.
The announcement, foreshadowed the day before by HHS Secretary Xavier Becerra, marks a profound shift in science policy and social values from former president Donald Trump to the three-month-old Biden administration.
The White House remained silent Friday about NIH's move. Yet rescinding the restrictions on federal support for fetal tissue research conforms to the oft-stated pledge by President Joe Biden and his top health advisers - largely in the context of trying to defeat the coronavirus pandemic - that they will place primacy on science.
Two years ago, the question of whether government money should continue to be invested in studies relying on cells from aborted fetuses became a major collision point between social conservatives and the nation's scientific community.
In 2019, Trump overrode the advice of his HHS secretary, Alex Azar, by blocking the use of NIH funds to purchase fetal tissue for use in government research labs. For outside researchers, the ban was less explicit, saying any application for a NIH grant or contract involving fetal tissue would need to be screened by a new ethics advisory board, if the application was deemed worthy on its scientific merits.
The Trump administration did not form the panel for about a year. When it did, most of its members strongly identified with opposition to abortion. The board convened once last summer before disbanding. It reviewed 14 proposals and recommended rejecting all but one.
Behind the scenes, senior figures at NIH were known to disagree with the two-part restrictions, though they did not speak against them publicly.
NIH's actions Friday rescind the central parts of the Trump-era policy but do not entirely restore rules to the way they had been. The 2019 rules compelled academic and other outside scientists applying for grants or contracts to start including extensive justification of why human fetal tissue was needed for the work and why other research methods were inadequate. Scientists complained the justification took up too much space in grant proposals and contract applications with a length limit.
Friday's changes leave in place the requirement for that extensive justification, according to proponents and opponents of the rewritten rules.
The policy change immediately flipped the politics surrounding federal fetal tissue policy. Scientists and their representatives who decried the Trump administration rules are celebrating their demise. Meanwhile, abortion opponents who advocated the last administration's policy Friday are bemoaning that it is now abandoned.
"The scientific community appreciates that the Biden administration is lifting the arbitrary restrictions on promising biomedical research using human fetal tissue," Christine Mummery, president of the International Society for Stem Cell Research, said in a statement. Saying that NIH should be "insulated from political interference," Mummery said Friday's actions were a "return to evidence-based policymaking."
Lawrence S.B. Goldstein, a senior researcher at the University of California at San Diego and the only member of the Trump administration's ethics advisory board who has used fetal tissue in his work, said, "It's an unqualified win. . . . Hopefully, all those great research projects will come back that were killed by the ethics advisory board."
Goldstein said, "I hope we are not going to enter an era of policy yo-yo as we change administrations," but he added, "For the moment, I am happy."
On the other hand, Tom McClusky, president of March for Life Action, an antiabortion advocacy group, said the research that the new rules will help permit "is a gross violation of human dignity. . . . The government has no business creating a marketplace for aborted baby body parts."
Tony Perkins, president of the like-mindedFamily Research Council, condemned Becerra as "a fanatical advocate for abortion."
David Prentice, another member of the ethics advisory board and research director of the antiabortion Charlotte Lozier Institute, said, "It's a sad turn of events. . . . Now, taxpayer funds will be used for research that is poor ethics and poor science. We've just backed away from any sort of ethical consideration around this issue."
Prentice said that under Friday's turnabout, "We're trafficking in aborted fetal tissue for antiquated experiments." He contended that fetal tissue is less effective in research than adult stem cells, artificially grown groups of cells known as organoids and other material not derived from aborted fetuses.
Asked whether Lozier or other organizations opposed to fetal tissue research might legally challenge the Biden rules, Prentice said, "At this point, we are reviewing all possible avenues."
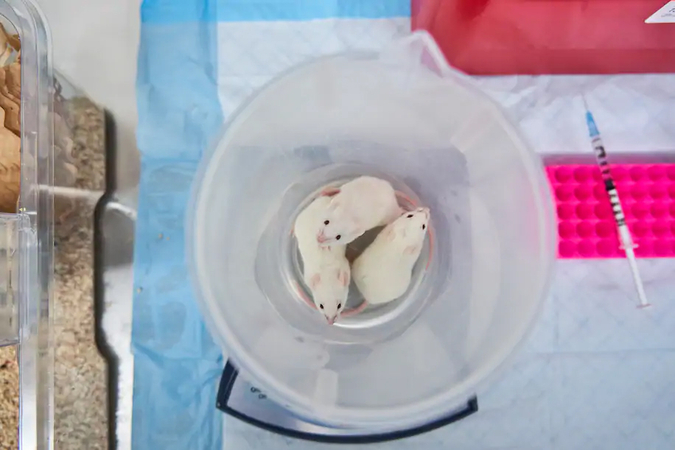
The role of fetal tissue in biomedical research extends to the 1950s, when Swedish researchers developed a polio vaccine by using fetal cells. In the late 1980s, scientists developed the technique of breeding mice with deficient immune systems and transplanting into them small amounts of immune system tissue from aborted fetuses.
These "humanized" mice grow the equivalent of a human immune system. They have become crucial lab animals in studying several major diseases, including therapies for HIV, cancers, neurological problems, sickle cell disease and eye disorders. NIH, by far the largest funding source for biomedical research in the United States, has paid for most of this work. Last year, a federal researcher was blocked from using fetal tissue to try to develop treatment for covid-19, the disease caused by the coronavirus.
Friday's updated NIH guidance for outside researchers said HHS and NIH "will not convene another NIH Human Fetal Tissue Research Ethics Advisory Board." It said researchers applying for federal grants or contracts involving work with fetal tissue still must obtain consent from the tissue's donor, may not pay for such tissue and must follow all other federal, state or university requirements.
For scientific studies undertaken by government employees, a one-paragraph email from Michael M. Gottesman, NIH's deputy director for "intramural" - or internal - research said guidelines "will return to their previous state." Human fetal tissue, the email said, "may again be used" in NIH's laboratories.
Work that requires new purchase of fetal tissue "from all previously-approved sources may be conducted," the notice said. Projects approved for use of fetal tissue before June 2019, when Trump changed the rules "will be reinstated without further review."
The Biden administration has been making piecemeal changes to conservative health-related policies of its predecessors.
In February, federal health officials rescinded a Trump administration policy allowing states to ask for permission to require some low-income residents to work or prepare for jobs to qualify for Medicaid. Instead of cutting back on efforts to urge Americans to enroll in Affordable Care Act health plans, the current administration created an unprecedented extended sign-up period, then lengthened it from three months to six months.
Biden officials had not publicly discussed a pending change in the rules for fetal tissue research until Thursday. Testifying at a budget hearing on Capitol Hill, Becerra said an announcement from NIH was imminent, and he made clear the direction it would take, thought not the details.
"We believe that we have to do the research it takes to make sure that we are incorporating innovation and getting all of those types of treatments and therapies out there to the American people," Becerra said.

