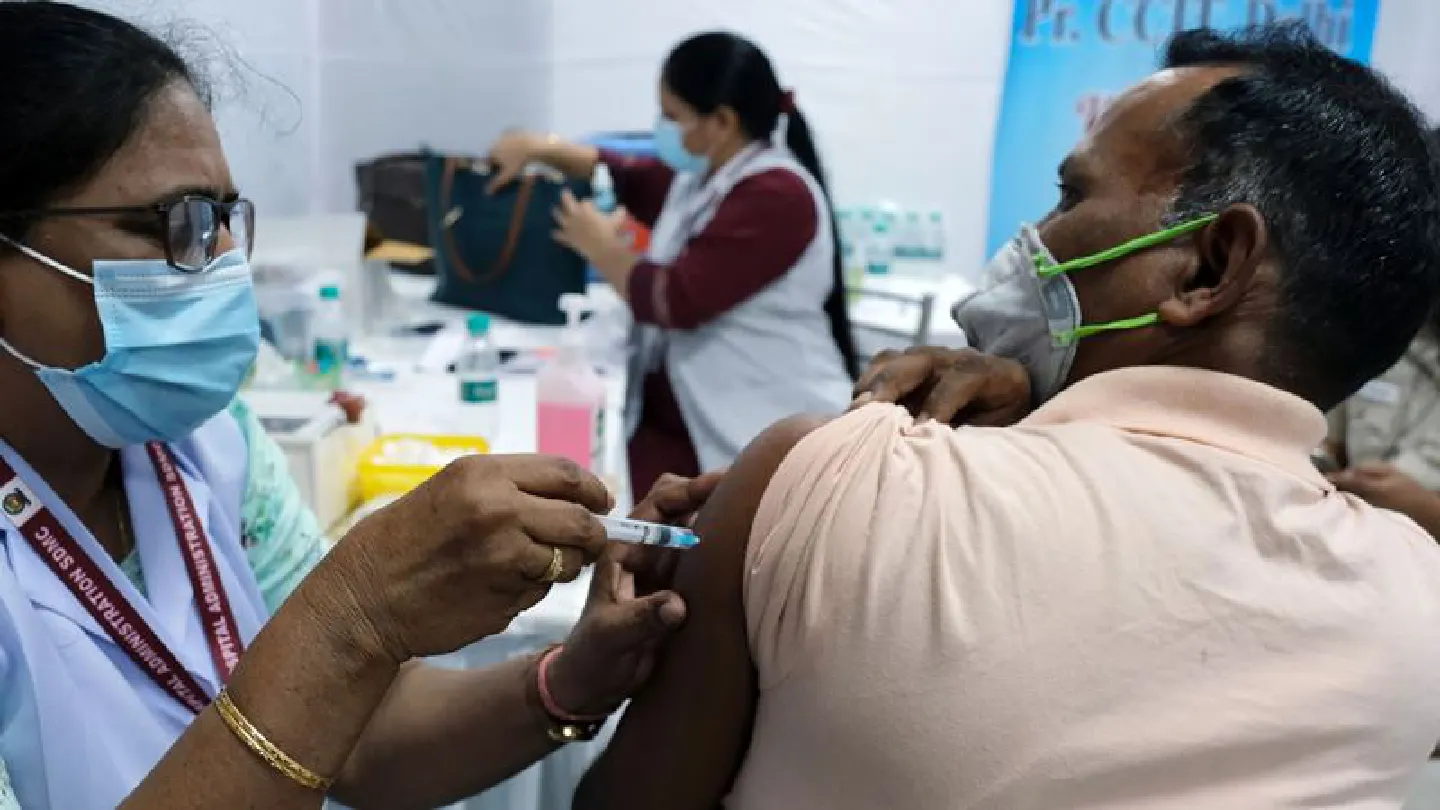
An Indian covid-19 shot showed 78% efficacy from interim data at preventing mild, moderate and severe reactions to the disease and worked against most variants of the virus as the South Asian nation struggles to contain a record surge across the country.
The inoculation known as Covaxin, a two-shot injection that uses an inactivated or dead form the virus, also showed 100% efficacy at preventing severe symptoms and hospitalizations against the coronavirus, vaccine maker Bharat Biotech International and the Indian Council for Medical Research, the co-developers, said in a joint statement on Wednesday.
The data from final stage trials that enrolled 25,800 participants has yet to be peer reviewed and a final analysis will be available from June, according to the statement.
"Covaxin works well against most variants of SARS-CoV-2," Balram Bhargava, the director general of the Indian Council of Medical Research, said in the statement, which added that several million doses of Covaxin had already been administered in the country. The medical agency also said in a Tweet that the vaccine "effectively neutralizes" a double mutant strain that has been detected and caused widespread concern in India.
The latest readings add to another interim statement last month that claimed the vaccine showed an 81% efficacy rate in those without prior infection after a second dose, adding to certainty over Covaxin after the shot was contentiously approved in January before it had completed Phase 3 testing. The vaccine was repeatedly marred by controversies almost as soon as work on its development was authorized last June. Those ranged from unrealistic government schedules for its release to sporadic reports of adverse reactions.
Now with India's health system on the verge of collapse as it hits record rates of covid infections and deaths, the government is pressuring the country's vaccine makers to quickly speed up their output amid a supply crunch. Bharat Biotech, which has been earmarked for a 650 million rupee ($8.6 million) government grant, said on Tuesday that it would try to scale up production of Covaxin to about 700 million doses on an annual basis and double its output by June and then push out nearly 100 million doses per month by September.
India has currently authorized three vaccines. Two of them from Astrazeneca Plc and Bharat Biotech are already in use, while the third -- Russia's Sputnik V -- was greenlit last week. The South Asian nation has also fast-tracked approval for foreign vaccines.
Despite India curbing vaccine exports this month as it attempts to get a grip on a new and overwhelming wave, Bharat Biotech said more than 60 countries have expressed interest in Covaxin, which "has quietly validated our efforts," Suchitra Ella, the company's joint managing director, said in the statement.
However, in March Brazil's health agency blocked the country's importation of 20 million Covaxin doses after inspectors said Bharat Biotech's facilities didn't meet requirements following an audit.

