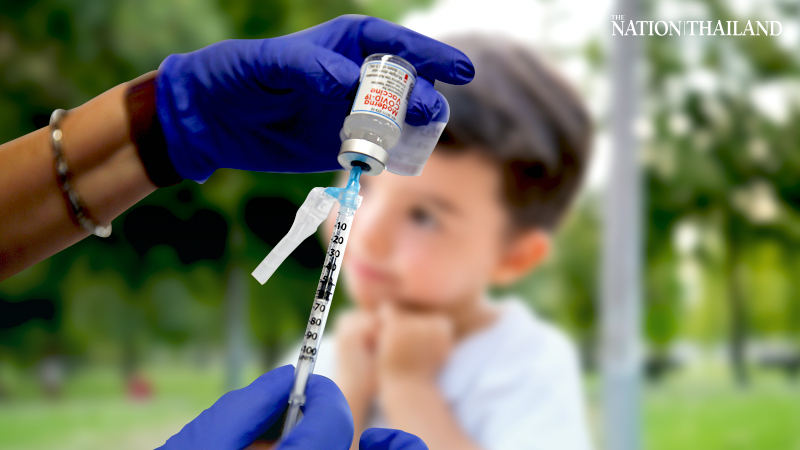
Moderna Inc. said the first children have been dosed in a clinical trial of its coronavirus vaccine in kids from six months to less than 12 years old.
The phase 2-phase 3 trial is being conducted in conjunction with the National Institute of Allergy and Infectious Diseases, the company said Tuesday in a statement. The study is expected to enroll approximately 6,750 participants in the U.S. and Canada.
The two-part study will look at three doses in the youngest children and two doses in older kids in its first phase. An interim analysis will be conducted to determine what dose will be used in the second part of the study. Kids who receive the vaccine will be followed for 12 months.
"This pediatric study will help us assess the potential safety and immunogenicity of our coronavirus vaccine candidate in this important younger age population," Moderna Chief Executive Officer Stephane Bancel said.
For adults, Moderna's vaccine was found to be safe and 94.1% effective at preventing covid-19, and it appears to work well across demographic groups and in people with underlying medical conditions. It is given in a two-dose regimen.
Moderna got emergency authorization for use of the shot in people ages 18 and older from the U.S. Food and Drug Administration in December. It has since become a key component of the immunization campaign in the U.S., with more than 52 million Moderna doses administered as of Monday, according to the Centers for Disease Control and Prevention.

